 
For 100% efficiency T c should equals to 0 K. Η = (T h - T c) / T h = 1 - T c / T h (1)Īs a consequence, to attain maximum efficiency - T c has to be as cold as possible. The thermodynamic efficiency of a heat machine working between two energy levels is defined in terms of absolute temperature and can be expressed as entropy can be produced but never destroyed.100% energy can not be transformed to work.Useful work must be derived from the energy that flows from the high level to the low level. The Second Law states that the entropy of the universe increases.įor energy to be available there must be a region with high energy level and a region with low energy level. The Second Law is concerned with Entropy (S) which is produced by all processes and associated with the loss of ability to do work. 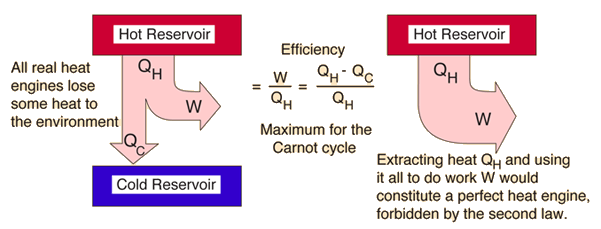 
"No machine whose working fluid undergoes a cycle can absorb heat from one system, reject heat to another system and produce no other effect"īoth statements of the Second Law constrains the First Law of Thermodynamics by identifying that energy goes downhill. 
"No (heat) engine whose working fluid undergoes a cycle can absorb heat from a single reservoir, deliver an equivalent amount of work, and deliver no other effect" 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
